In other diseases, including such diverse entities as primary CNS neoplasms and various movement disorders, this relationship has not been confirmed but appears likely in the light of rapidly accumulating clinical, epidemiological, and biochemical data. Relationships between endogenous and exogenous sex hormones and many neurological conditions, such as migraine, stroke, and chorea, are well established. Sex hormones influence a broad spectrum of normal and abnormal neurological functions. Schipper, in Neurology and General Medicine (Fourth Edition), 2008 CONCLUDING COMMENTS Collectively, these direct and indirect actions of sex steroids on bone and calcium metabolism are critical for normal bone mass acquisition and maintenance. In addition, sex steroids also affect bone turnover indirectly, via extraskeletal effects on calcium homeostasis. Conversely, androgens also have important skeletal effects in both sexes. Estrogen likely has a significant effect on bone not just in females, but also in males. While there are several candidate factors for mediating the autocrine and paracrine effects of estrogen and, to a lessor extent, androgens, on osteoclasts and osteoblasts, the precise mechanism of direct sex steroid effects on the skeleton remains to be clearly defined. These include effects on longitudinal growth at the time of puberty, the acquisition of peak bone mass, and the regulation of bone turnover in adults. Sex steroids clearly have major effects on all aspects of bone metabolism. KHOSLA M.D., in Dynamics of Bone and Cartilage Metabolism (Second Edition), 2006 VII. Aromatase also has protective effects, probably through its action to increase estrogen and to decrease testosterone.ĭAVID G. In contrast, testosterone, in the same model, has the opposite effect. 79 Estrogen, through the activation of ERβ, confers protection after ischemia and reperfusion in murine models of myocardial infarction. The modulatory actions of estrogen on Ca 2+ channels might be responsible for sex-based differences in repolarization of the heart, such as the faster resting heart rate of women, as well as the increased propensity of women to have prolonged QT syndrome. Gender differences exist in cardiac electrophysiologic function. In addition to sex steroid hormone stimulation of nuclear or nonnuclear receptors, sex steroid hormone receptors could also induce rapid signaling of growth factor pathways in the absence of ligands. In men, aromatase-mediated conversion of testosterone to estrogen maintains normal vascular tone. Estrogen’s vasodilatory effect might explain the lower systolic blood pressures of premenopausal women when compared with age-matched men. One such example is stimulation of vascular endothelial nitric oxide synthase to mediate vascular dilation. 78 Sex steroid hormones can activate rapid signaling pathways without changing gene expression ( Fig.
These cell-specific coactivator and corepressor proteins are known as estrogen-related receptors. They interact with many different coregulators to confer tissue and temporal specificity in their transcriptional actions. Cardiomyocytes express nuclear steroid hormone receptors that modulate gene expression and nonnuclear receptors for the nongenomic effects of sex steroid hormones. Estradiol metabolism also takes place in vascular smooth muscle cells, cardiac fibroblasts, endothelial cells, and cardiomyocytes. 77Į2 is derived from testosterone and is primarily metabolized in the liver to form hydroxyestradiols, catecholestradiols, and methoxyestradiols. Cardiomyocytes are not only targets for the action of sex steroid hormones, but they are also the source of synthesis and the site of metabolism of these hormones. Sex steroid hormones interact with their receptors to affect postsynaptic target cell responses and to influence presynaptic sympathoadrenergic function.
Progesterone and testosterone bind and act on their respective progesterone receptors and androgen receptors in the heart. Progesterone and testosterone (two other sex steroid hormones) and the enzyme aromatase, which converts testosterone to estrogen, are much less well investigated. They bind and act on the two subtypes of estrogen receptors (ERs) in the heart: ERα and ERβ. The most extensively studied sex steroid hormones are estradiol-17β (E2) and its bioactive metabolites. Indeed, healthy premenopausal women exhibit a lower cardiovascular risk compared to men, which suggests a mechanism for sex hormones in the modulation of cardiac function. The gender dimorphism in heart function and its adaptive responses to injury and disease states are partly mediated by sex steroid hormones.
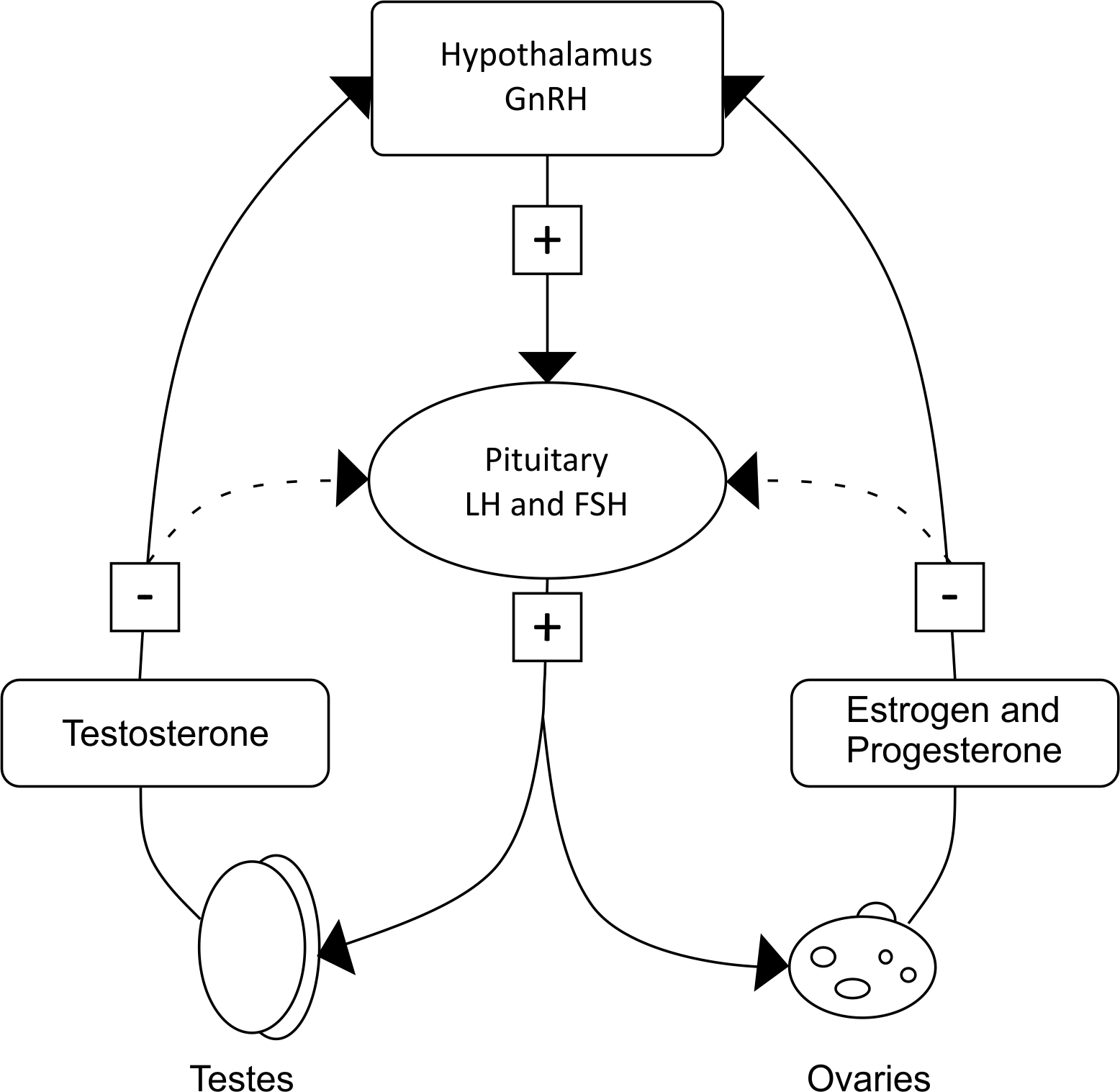
Gropper MD, PhD, in Miller's Anesthesia, 2020 Sex Steroid Hormones and the HeartĬardiac contractility is more intense in premenopausal women than in age-matched men, and withdrawal of hormone replacement therapy in postmenopausal women leads to a reduction in cardiac contractile function.
